Scientists exploring new treatments for multiple sclerosis say an experimental approach originally developed to fight cancer could offer a promising path forward. Early research into CAR-T Therapy suggests the powerful immune-cell treatment may help target the underlying immune dysfunction that drives the disease.
Multiple sclerosis, often referred to as MS, is a chronic autoimmune condition that affects the central nervous system. The disease occurs when the immune system mistakenly attacks myelin, the protective layer surrounding nerve fibers in the brain and spinal cord. As the damage spreads, communication between the brain and the rest of the body becomes disrupted, leading to a wide range of neurological symptoms.
Researchers now believe that CAR-T Therapy may offer a way to reset parts of the immune system responsible for these attacks. While the concept remains experimental for MS, small clinical trials underway in the United States and Europe are beginning to explore whether the therapy can slow or halt disease progression.
Understanding Multiple Sclerosis and Immune System Damage
Multiple sclerosis affects nearly three million people worldwide and is one of the most common neurological diseases among young adults. Symptoms can vary widely, ranging from fatigue and vision problems to difficulties with balance, walking, and cognitive function.
The condition develops when immune cells attack the myelin sheath that protects nerve fibers. Over time, this damage leads to inflammation and scarring in the brain and spinal cord. When the protective coating deteriorates, nerve signals slow down or fail to travel properly through the nervous system.
Doctors have developed several medications that can reduce inflammation and slow the immune system’s attack on myelin. However, many treatments do not completely stop the disease, and patients may still experience gradual neurological decline.
Because of these limitations, scientists continue searching for therapies capable of addressing the root immune problem rather than only managing symptoms. One of the most intriguing possibilities emerging from recent medical research involves CAR-T Therapy.
Why Researchers Are Studying CAR-T Therapy for MS
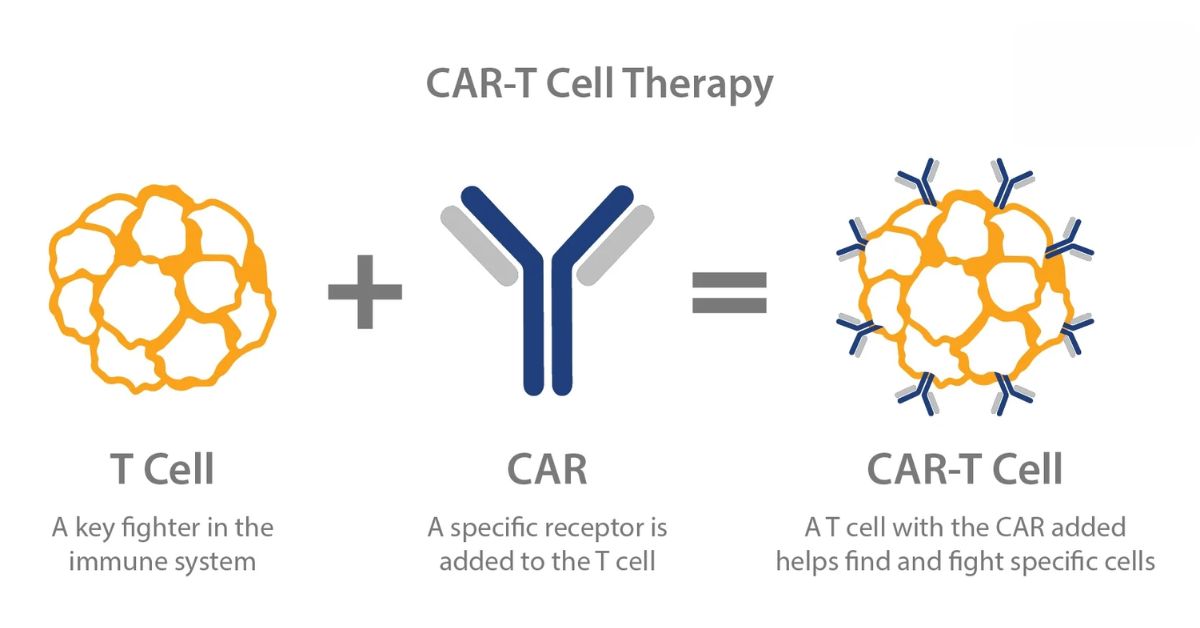
CAR-T Therapy was originally designed as a breakthrough treatment for certain cancers, particularly blood cancers such as leukemia and lymphoma. The therapy uses a patient’s own immune cells and modifies them so they can recognize and destroy specific targets inside the body.
In cancer treatment, those targets are malignant B cells. In autoimmune diseases like multiple sclerosis, researchers suspect similar immune cells may be responsible for damaging the nervous system.
Scientists studying CAR-T Therapy believe the treatment could remove the specific immune cells driving the disease process. By eliminating those cells, the immune system might reset itself and stop attacking the body’s own tissues.
Traditional MS drugs can suppress immune activity, but they do not always reach immune cells that hide within the brain and spinal cord. Researchers hope CAR-T Therapy may overcome this limitation by seeking out and destroying those harmful cells wherever they exist.
Although the approach is still in early stages, several academic medical centers have launched clinical trials to examine whether this strategy could change how autoimmune diseases are treated.
How CAR-T Therapy Works Inside the Body
The process behind CAR-T Therapy begins with collecting a sample of a patient’s T cells, which are a type of immune cell responsible for fighting infections and abnormal cells.
Doctors extract these cells from the patient’s blood and send them to a laboratory. There, scientists genetically modify the cells so they produce special receptors known as chimeric antigen receptors. These receptors allow the T cells to recognize a specific target on other immune cells.
Once modified, the engineered T cells are multiplied in large numbers before being infused back into the patient’s bloodstream.
After entering the body, the modified cells begin searching for immune cells carrying the targeted marker. When they find those cells, they attach to them and destroy them.
In cancer treatment, the goal is to eliminate malignant cells. In autoimmune diseases like multiple sclerosis, the goal is to remove immune cells that mistakenly attack the nervous system.
Researchers studying CAR-T Therapy believe that eliminating these destructive immune cells could potentially stop the autoimmune process responsible for MS symptoms.
Early Clinical Trials Show Encouraging Signals
Several early studies are currently underway to determine whether CAR-T Therapy can safely treat multiple sclerosis. These trials involve relatively small numbers of patients but are closely monitored by neurologists and immunology experts.
Some of the most closely watched research programs are being conducted at major medical institutions across the United States. The goal of these trials is not only to determine whether the therapy is safe but also to observe whether it produces measurable improvements in symptoms or disease progression.
Preliminary observations have shown that some patients receiving CAR-T Therapy experienced reduced signs of immune activity linked to multiple sclerosis. Researchers are also studying whether the treatment can prevent new lesions from forming in the brain, which is a common sign of disease progression.
While these findings remain early, scientists say the results are encouraging enough to justify further investigation. Larger trials will be required to determine whether the therapy can become a reliable treatment option.
A Patient’s Experience With Experimental Treatment
For some patients living with multiple sclerosis, the opportunity to participate in a clinical trial represents a chance to explore new possibilities after years of limited treatment options.
One participant in an early study had lived with MS symptoms for more than a decade. Fatigue, balance issues, and vision disturbances had gradually worsened despite trying several approved medications.
After enrolling in a clinical trial testing CAR-T Therapy, the patient received a single infusion of modified immune cells. Doctors monitored the patient carefully for months following the procedure.
Although the patient continued to experience some mobility limitations, doctors observed signs that disease activity had slowed. The patient also reported gradual improvements in strength and daily functioning.
Researchers caution that individual patient experiences cannot determine whether a treatment truly works. However, stories like these highlight why many scientists are interested in exploring the potential of CAR-T Therapy for autoimmune diseases.
Experts Urge Caution as Studies Continue
Despite the excitement surrounding early research, medical experts emphasize that CAR-T Therapy remains experimental in the context of multiple sclerosis.
Clinical trials must confirm that the therapy is both safe and effective before it can be approved for widespread use. That process often takes many years and requires extensive testing.
Some neurologists also point out that multiple sclerosis is a complex disease involving both immune activity and long-term nerve damage. Even if CAR-T Therapy successfully removes harmful immune cells, it may not repair damage that has already occurred in the nervous system.
Researchers therefore believe future treatments may need to combine immune system therapies with approaches that repair or regenerate damaged nerve cells.
For now, scientists continue studying how CAR-T Therapy interacts with the immune system in people living with MS.
Risks and Safety Concerns Researchers Are Monitoring
While CAR-T Therapy has shown remarkable results in cancer treatment, the therapy can also produce serious side effects.
One of the most well-known complications is cytokine release syndrome, a condition in which the immune system reacts aggressively after modified cells are introduced into the body. Symptoms can include fever, low blood pressure, and severe inflammation.
Another potential complication is neurological toxicity, which can cause confusion, headaches, or seizures in rare cases.
Because multiple sclerosis already affects the brain and nervous system, researchers studying CAR-T Therapy for MS are monitoring patients carefully for these complications.
Clinical trials include strict safety protocols designed to detect side effects early and provide treatment if complications arise.
Doctors involved in these studies emphasize that understanding the safety profile of the therapy is just as important as measuring its potential benefits.
The Future of CAR-T Therapy in Autoimmune Diseases
Although research is still in its early stages, scientists believe CAR-T Therapy could represent a major shift in how autoimmune diseases are treated.
If the therapy proves successful for multiple sclerosis, researchers say it could potentially be adapted for other conditions in which the immune system attacks healthy tissue.
Diseases such as lupus, rheumatoid arthritis, and myasthenia gravis are also being studied as possible candidates for immune-cell therapies.
For patients living with multiple sclerosis, the possibility of a treatment capable of stopping the disease at its source offers a powerful sense of hope.
Even if the therapy ultimately becomes only one part of a broader treatment strategy, scientists believe the research surrounding CAR-T Therapy is opening new doors in the fight against autoimmune diseases.
As clinical trials continue, researchers remain focused on understanding how the therapy works, how safe it is, and whether it can deliver lasting benefits for patients living with multiple sclerosis.